COMPANY
Go Home
-
Vision & Mission Statement
Vision & Mission Statement
-
Value
Striving for people’s happiness,
Contributing to enhancement of public health & welfare -
Creativity
Introducing superior products quickly to market, Developing products with high social demand
-
Public Benefit
Undertaking research projects to promote healthy lives of people
Vision & Mission Statement 1.
- ValueStriving for people’s happiness,
Contributing to enhancement of public health & welfare - Contributed to early testing of HCV & HIV
- Contributed to new health technology research and development (Please refer to
reimbursement/non-reimbursement notice by Ministry of Health and Welfare)
- Contributed to transformation of social perceptions regarding infectious diseases
- Contributed to development of innovative testing system for infection management
Close PopUp
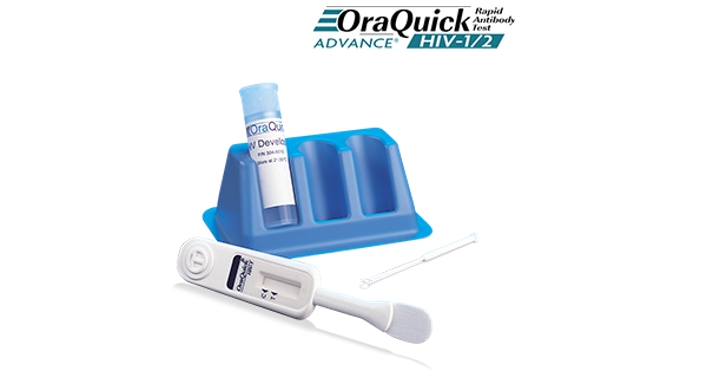
- Rapid Point-of-Care HIV Antibody Test
- Health Insurance Service Reimbursement Item
- HCV Antibody Test
- Health Insurance Service Reimbursement Item
Vision & Mission Statement 2.
- CreativityIntroducing superior products quickly to market,
Developing products with high social demand - Introducing superior products quickly to market, Developing products with high social demand
- Established online business via eSTIM
- Developed Oraquick Autotransmission system
Close PopUp
- QraQuick Autotransmission System

- Diagnostic Kit & Smartphone Stand
Vision & Mission Statement 3.
- Public BenefitUndertaking research projects to
promote healthy lives of people - Completed academic research project offered by Korea Disease Control and Prevention Agency (KDCPA)
[Project Title: “Development and Simulation of Routine HIV Testing System in Dental Clinic”]
- Conducted business in collaboration with Seoul Association of University, Research Institute and Industry (SAURI)
[Project Title: “Supporting Development of Techniques for Patented Technology Commercialization”]
- Founded “The Korean Academy of Dental Clinic Infection
Close Popup
Close PopUp